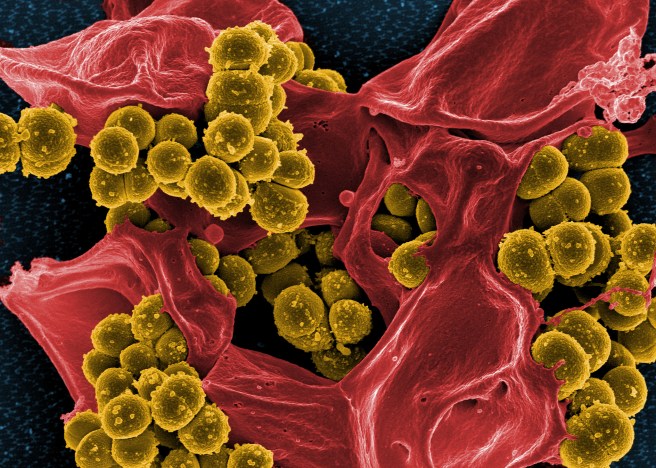
We hear a lot about toxins in the news these days. Specifically, the hidden toxins lurking in the food we eat, the household products we use, the air we breathe and why we need to go on a juice cleanse to detox our bodies, lose weight and feel great!
But right now, let’s ignore those exaggerations and pseudoscience (because that’s a lengthy post in and of itself) and talk about real toxins. Real bacterial toxins. These toxins are proteins made and secreted by bacteria that help them establish an infection and cause disease. Staphylococcus aureus, commonly known as staph, is one species of bacteria that deploys a large and diverse arsenal of toxins. Most people carry staph bacteria asymptomatically on their skin and in their noses. In certain individuals, such as those with a weakened immune system, the bacteria can cause a wide spectrum of diseases from minor skin and soft tissue infections to life-threatening pneumonia and bloodstream infections. A key component of the bacteria’s survival strategy are the toxins that damage tissues and attack immune cells to interfere with the host’s defense system. Toxins are also responsible for disease symptoms such as the skin lesions commonly seen in patients with a staph infection.
Given the important role that toxins play in establishing and maintaining an infection, it would be logical to assume that the more toxins a bacteria produces, the more severe the infection. Until recently, that was the prevailing belief in the research community. Continue reading


